




[Text en: Proteine]
Proteins are important building blocks of nature. These highly complex macromolecules are formed out of surprisingly few different amino acids. Proteins are specialists, always serving a very specific function in a cell, be it a cytochrome performing electron transport, or a channel protein, which - depending on specific environmental parameters such as pH or temperature - transports a specific type of ion - and only this type of ion! - through the otherwise impermeable cell membrane.
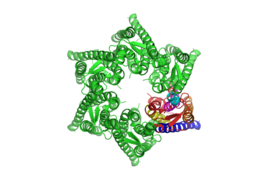
For example, the human Connexin 26 (hCx26) forms a channel to transport Ca2+-ions from one cell to the next. Each cell builds hemi-channel of hCx26 (see Figs. 1 and 2) into its membrane. If the two hemi-channels dock and open, Ca2+-ions are handed from one cell to the next.
Upon opening of the channel, the protein changes its molecular conformation, effectively widening its central pore and allowing small atoms to pass through. Often, amino acids with charged side-chains are exposed or buried by the conformational change, making the opening specific for ions. The experimental confirmation of these conformational changes of a protein's structure is one of the big challenges in protein science. Protein structures are usually analyzed with atomic force microscopy, x-ray cristallography, and electron force microscopy. However, the preparations required for these methods are often uncompatible with the specific environmental parameters causing protic conformational changes.
At HOT, we used high-precision confocal Raman microscopy for in situ analysis of these protein conformations. Without fixing or cristallizing the proteins, it is possible to record and compare the Raman spectra of the protein in different conformations. For example, we were able to confirm the assumption that the complete closure of the aforementioned hCx26 at temperatures below 23°C involves a rotation of the extracellular loop attached to the first transmembrane helix and possibly also the fourth transmembrane helix. In particular, we found a direct indicator for a closed hemi-channel in the Raman spectrum itself.
See also
A.-K. Kniggendorf, M. Meinhardt-Wollweber, X. Yuan, B. Roth, A. Seifert, N. Fertig, and C. Zeilinger: Temperature-sensitive gating of hCx26: high-resolution Raman spectroscopy sheds light on conformational changes
2014. Biomedical Optics Express 5 (7) doi: 10.1364/BOE.5.002054
